39 open label meaning
Open-label extension studies: do they provide meaningful ... - PubMed Open-label extension studies do have a legitimate but limited place in the clinical development of new medicines. The negative perceptions about these studies have arisen because of perversion of acceptable rationales for this type of study and a failure to recognise (or disclose) the limitations resulting from the inherent weaknesses in their design. Open-Label Trial - an overview | ScienceDirect Topics Open-label trials of desipramine, tranylcypromine, reboxetine, ... Although there is some debate over what "triple-blind" means, we define it as the patient, the researchers conducting the study, and anyone analyzing the results (e.g., sponsor) do not know which subjects belong to which study arms. Triple-blinding may be useful when ...
Meaning of "Open Label Pilot study"..?? - SAS Support Communities Open label is a term used to describe the situation when both the researcher and the participant in a research study know the treatment the participant is receiving. Open-label is the opposite of double-blind when neither the researcher nor the participant knows what treatment the participant is receiving. Pilot study means a smaller version of a ...
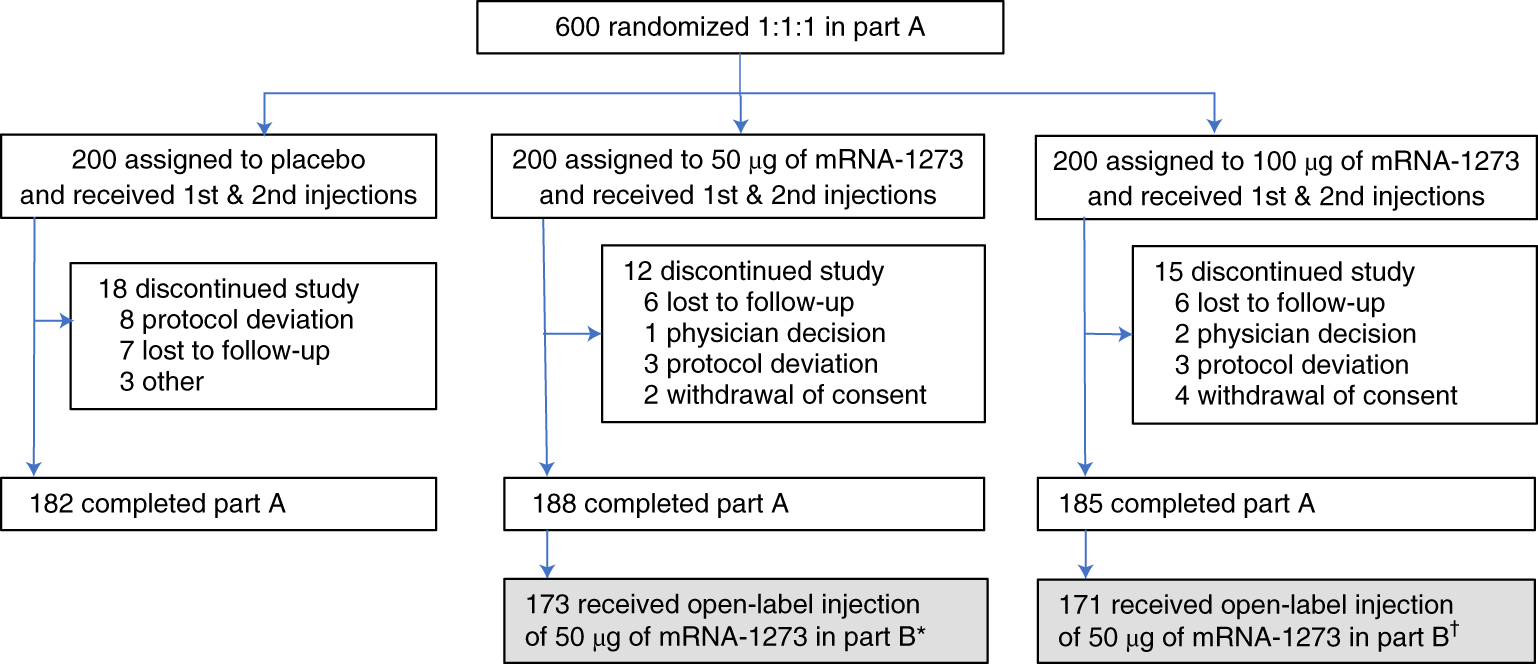
Open label meaning
National Center for Biotechnology Information National Center for Biotechnology Information Here's What 10 Symbols on Cosmetics Labels Mean | Mental Floss It turns out that these icons are a great way to get to know the cosmetics that serve to make us more presentable human beings. 1. PAO (Period After Opening) The little open jar icon is the PAO ... National Cancer Institute NCI's Dictionary of Cancer Terms provides easy-to-understand definitions for words and phrases related to cancer and medicine.
Open label meaning. What is an open label trial? | The BMJ Open label trials are sometimes referred to as "non-masked" or "unblinded." If the trial is a non-pharmacological study, such as a trial of devices, or psychological and physical treatments, it may be referred to simply as "open." After recruitment to the trial, the participants were allocated to treatment using block randomisation. Label definition and meaning | Collins English Dictionary Label definition: A label is a piece of paper or plastic that is attached to an object in order to give... | Meaning, pronunciation, translations and examples A Multi-center, Open-label, Randomized, Parallel Group Study to ... This study is an open-label randomized trial, meaning that you and your study doctor will be aware of the dosage of study drug that you will be given. Randomization means that you will be selected by chance, like tossing a coin or rolling a dice. ... This is a Phase I/II, multi-center, open-label, randomized, parallel group, pharmacokinetic ... Open-label Definition & Meaning - Merriam-Webster Definition of open-label. : being or relating to a clinical trial in which the treatment given to each subject is not concealed from either the researchers or the subject an open-label multicenter study — compare double-blind, single-blind.
The Personal Care Association :: Understanding the Label - Cosmetics Europe The nominal net. Any warnings that might be necessary on how to use the product safely. A "date of minimum durability" ("best used before the end of") or a "period after opening" to show for how long the product may be kept or used. What the product is (if not obvious from its appearance). A reference (batch number) for product identification. Open-label study | definition of open-label study by Medical dictionary open-label study a study in which there is no blinding of treatments. Farlex Partner Medical Dictionary © Farlex 2012 open-label study A clinical study in which the patients/subjects and investigators know which product each patient/subject is receiving, which is the opposite of a blinded study. Segen's Medical Dictionary. © 2012 Farlex, Inc. Open-label - definition of open-label by The Free Dictionary o·pen-la·bel. (ō′pən-lā′bəl) adj. Of or related to a clinical trial in which both the researcher and the participant know whether an administered treatment is an experimental drug or a placebo. American Heritage® Dictionary of the English Language, Fifth Edition. Open letter Definition & Meaning - Merriam-Webster Definition of open letter : a published letter of protest or appeal usually addressed to an individual but intended for the general public Examples of open letter in a Sentence In an open letter to the company, a citizens' group implored company executives to reconsider their decision to close the factory.
LABEL | meaning in the Cambridge English Dictionary label definition: 1. a piece of paper or other material that gives you information about the object it is attached…. Learn more. Medical Definition of Open-label - MedicineNet Open-label: A term used to describe the situation when both the researcher and the participant in a research study know the treatment the participant is receiving. Open-label is the opposite of double-blind when neither the researcher nor the participant knows what treatment the participant is receiving. Why an open banking white-label solution? | Klarna International Open banking customers who use the Klarna branded API use a system built on the same technical foundations as our white label solution. That means we know it works, and it works well. Smoooth White Label Banking Software from Start to Finish. When choosing a white label solution, the first thing to check for is the provider's experience. White labelling benefits from strong relationships that providers, like Klarna, have with banks across multiple countries. What Is a Volume Label of a Drive? - Lifewire A volume label, sometimes called a volume name, is a unique name assigned to a hard drive, disc, or other media. It's not required in Windows, but it's often useful to give a name to a drive to help identify its use in the future. A drive's volume label can be changed at any time, but is usually set during the formatting of the drive.
Open-label trial - Wikipedia An open-label trial, or open trial, is a type of clinical trial in which information is not withheld from trial participants. In particular, both the researchers and participants know which treatment is being administered.
Understanding Clinical Trial Terminology: What is an Open Label ... Open-label trials can be used to compare treatments or gather additional information about the long-term effects in the intended patient population. In some instances, patients who complete one clinical trial may be eligible to continue in an open-label extension study where all participants are eligible to receive active treatment for an extended period of time.
Effects of open-label placebos in clinical trials: a ... - Nature Open-label placebos (OLPs) are placebos without deception in the sense that patients know that they are receiving a placebo. The objective of our study is to systematically review and analyze the ...
Open-Label Trial | NIH - HIV.gov A type of clinical trial. In open-label trials, both the researchers and participants know which drug (or other intervention) is being given to participants.
What Symbols on Cosmetic Labels Mean - Consolidated Label Below are symbols that can be seen on cosmetic labels in the United States and European Union, and what they mean. Period After Opening (PAO): The open jar symbol tells you how long the product will stay good after it's been opened in months and/or years.
Off-label use - Wikipedia Off-label use is the use of pharmaceutical drugs for an unapproved indication or in an unapproved age group, dosage, or route of administration. Both prescription drugs and over-the-counter drugs (OTCs) can be used in off-label ways, although most studies of off-label use focus on prescription drugs.. Off-label use is very common and generally legal unless it violates ethical guidelines or ...
Understanding Clinical Trial Terminology: What is a Long-Term Extension ... An OLE study is a clinical trial that typically enrolls participants of a previous clinical trial and is designed to gather the long-term safety and tolerability data on a potential new medicine beyond the time period of the main study.
Open-label-Studie - DocCheck Flexikon Definition. Als Open-label-Studie bezeichnet man eine klinische Studie mit einem Studiendesign, bei dem sowohl die Probanden als auch der Prüfarzt über den verabreichten Wirkstoff in Kenntnis gesetzt werden. Dieses "entblindete" Studiendesign stellt das Gegenstück einer Einfachblindstudie und Doppelblindstudie dar. Fachgebiete: Ivan Lechner.
Cute Sticky Note Student Message Sticker N Times Memo Pad Scrapbooking School Label Stationery - Buy Sticky Notes Daily Memo Book Creative Message Memo,Custom Notepad Memo Pad Sticky Notes Many Bases ...
Medical Definition of Open-label trial - MedicineNet Privacy & Trust Info Open-label trial: A clinical trial in which researchers and participants know which drug or vaccine is being administered. CONTINUE SCROLLING OR CLICK HERE Health Solutions From Our Sponsors Penis Curved When Erect Could I have CAD? Treat Bent Fingers Treat HR+, HER2- MBC Tired of Dandruff? Life with Cancer
National Cancer Institute NCI's Dictionary of Cancer Terms provides easy-to-understand definitions for words and phrases related to cancer and medicine.
Here's What 10 Symbols on Cosmetics Labels Mean | Mental Floss It turns out that these icons are a great way to get to know the cosmetics that serve to make us more presentable human beings. 1. PAO (Period After Opening) The little open jar icon is the PAO ...
National Center for Biotechnology Information National Center for Biotechnology Information




























Post a Comment for "39 open label meaning"